Our research spans a broad range of initiatives in gastrointestinal endoscopy, organized across three core areas: Artificial Intelligence in Endoscopy, Randomized Clinical Trials, and Registries and Cohort Studies. Explore below to learn more about our ongoing projects.
Research
Cohort Studies
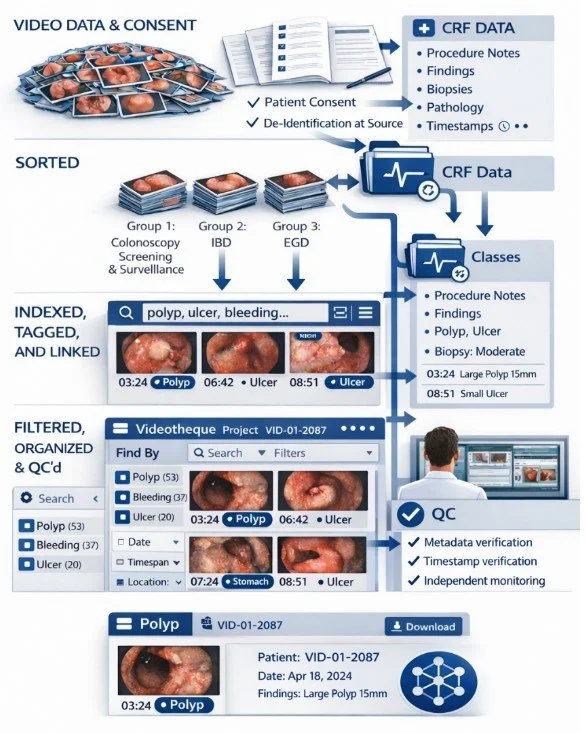
VIDEOTHEQUE
The Videotheque is a prospective, continuously growing video databank of gastrointestinal endoscopic procedures established at the Centre Hospitalier de l'Université de Montréal (CHUM). Unlike most existing endoscopy datasets, which are retrospective, image-based, or assembled without structured clinical context, the Videotheque was designed to capture full-length procedure recordings alongside richly annotated clinical, procedural, and histopathological data. Every enrolled procedure is documented in real time by dedicated research staff present in the procedure room, ensuring annotations reflect what was actually seen and done, not reconstructed from reports after the fact.
Each procedure is linked to hundreds of structured variables spanning patient demographics, procedural technique, lesion characteristics, optical diagnoses, and pathology results. Lesion-level annotations are timestamped directly to the corresponding video frames, creating a granular resource where every finding can be traced back to its precise moment in the recording , across colonoscopy, upper GI endoscopy, and IBD surveillance.
What makes the Vidéothèque particularly valuable for AI development is the quality and provenance of its labels. Because every annotation was prospectively assigned using standardised protocols, the data provides a trustworthy foundation for developing and validating computer vision models across endoscopic tasks, from polyp detection and optical diagnosis to disease activity scoring. Beyond AI, the databank supports clinical outcomes research through longitudinal follow-up, pathology correlation, and quality indicator tracking that retrospective records simply cannot provide.
AE-Registry
The AE Registry is a prospective, multicentre registry tracking real-world outcomes of advanced endoscopic procedures across Canada. The registry enrolls patients undergoing endoscopic mucosal resection (EMR), endoscopic submucosal dissection (ESD), deep dissection, radiofrequency ablation, and endoscopic myotomy, procedures that increasingly replace surgery for the treatment of complex gastrointestinal lesions.
At its core, the AE Registry is a quality improvement tool. By systematically collecting structured clinical data on technique, resection completeness, complications, and long-term oncologic outcomes, the registry allows participating centres to benchmark their practice, identify gaps, and implement targeted improvements. Data on endoscopist technique, instrument choice, and electrocautery settings are linked directly to patient outcomes, creating a feedback loop that drives better, safer endoscopy.
Beyond quality monitoring, the registry serves as a platform for outcomes research. With up to 20 years of follow-up per patient, the AE Registry is designed to answer long-term questions about recurrence, cancer-related mortality, and the factors that predict procedural success, evidence that cannot be generated from retrospective records alone.
Advanced Endoscopic Procedures Registry
Randomized Controlled Trials (RCT’s)
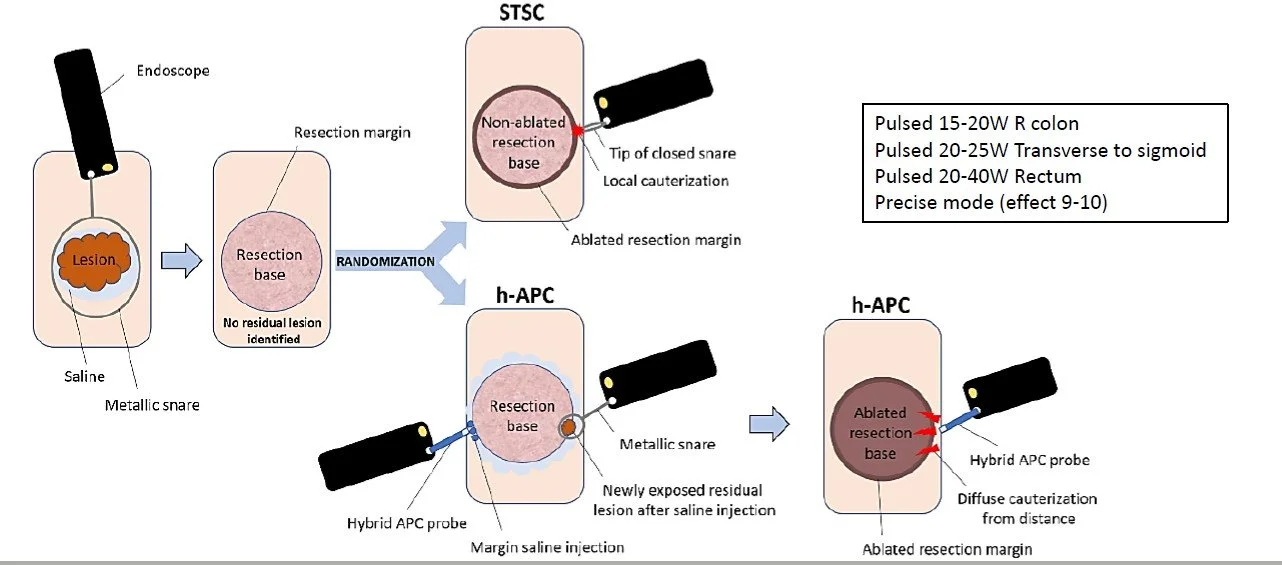
hAPC RCT
A multicenter RCT comparing two thermal ablation techniques after EMR of large colorectal polyps (≥20mm): hybrid Argon Plasma Coagulation (h-APC), which ablates both the margin and base, versus standard snare tip soft coagulation (STSC), which ablates the margin only. The primary outcome is histological recurrence at 6 months, across 848 patients at 13 sites in Canada, the USA, and Europe.
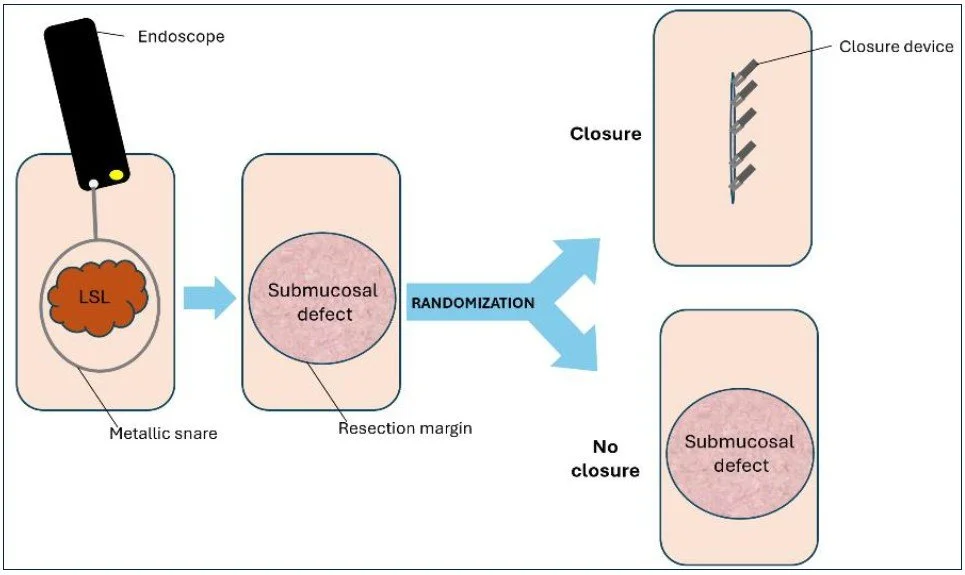
Closure RCT
A multicenter randomized controlled trial investigating whether complete defect closure following EMR of large colorectal polyps (≥20mm) reduces clinically significant adverse events — including delayed bleeding and perforation — compared to no closure. Using next-generation clip devices (MANTIS and X-Tack), this trial aims to achieve 100% closure rates across 686 patients at 12 sites in Canada and the United States.
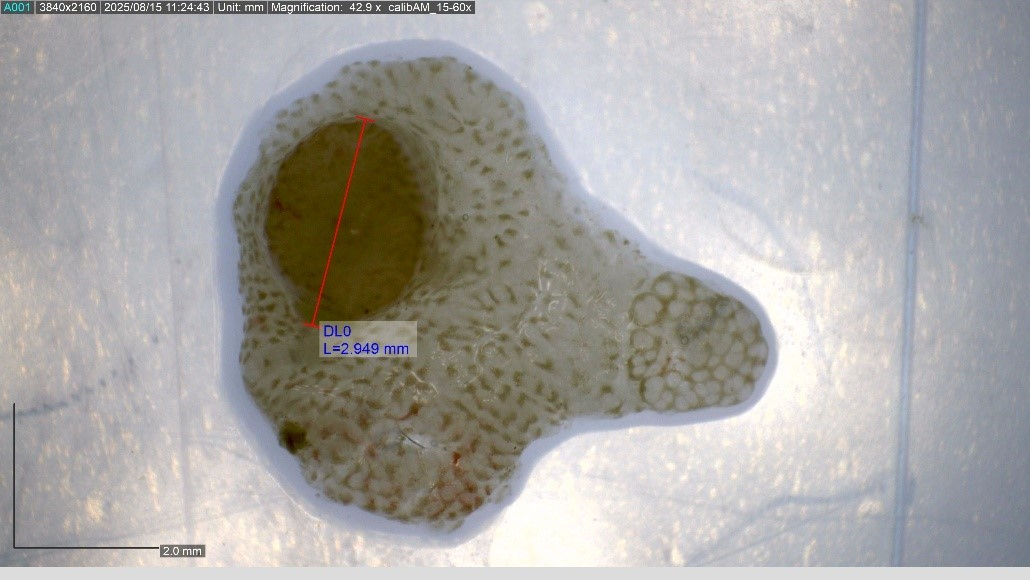
Polyp Sizing
Accurate sizing. Better decisions. Fewer missed cancers.
Accurate polyp size estimation is one of the most consequential, and most variable, measurements in endoscopy. Size determines surveillance intervals, resection strategy, and cancer risk classification, yet endoscopist visual estimation remains the standard in most centres despite well-documented inaccuracy.
At CEDIA, we systematically study and compare multiple sizing methods, including endoscopist visual estimation, caliper-based measurement, laser-assisted sizing tools, and microscopic measurement of resected specimens, evaluating their accuracy, reproducibility, and clinical impact. We are building a rigorous evidence base for which methods perform best, in which contexts, and for which lesion types.
Our work also explores how AI-based sizing tools can reduce measurement variability and improve consistency across endoscopists, with the goal of standardising this critical step and ensuring that every patient receives the right follow-up based on reliable data.
AI in Endoscopy
Building, testing, and advancing the next generation of endoscopy AI.
At CEDIA, we both develop and independently evaluate AI systems for gastrointestinal endoscopy. We benchmark commercially available AI tools against endoscopist performance and histopathology-confirmed ground truth, and apply the same rigour to open-source and foundation models, assessing their performance and potential for clinical adaptation.
Using the Videotheque as our primary resource, we develop and validate AI models across tasks ranging from polyp detection and optical diagnosis to disease activity scoring. We also collaborate with academic, clinical, and industry partners to co-develop and evaluate AI systems, contributing our dataset infrastructure and independent evaluation expertise to joint projects.
Our goal is simple: ensure that AI in endoscopy is built and tested the right way, rigorously, transparently, and with patients at the centre.
